|
4/1/2024 0 Comments Sp3d hybridization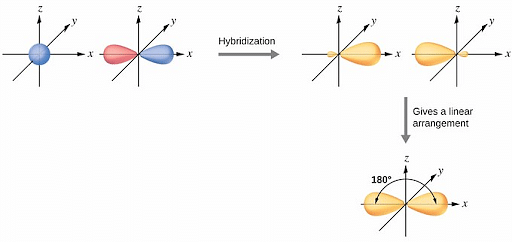 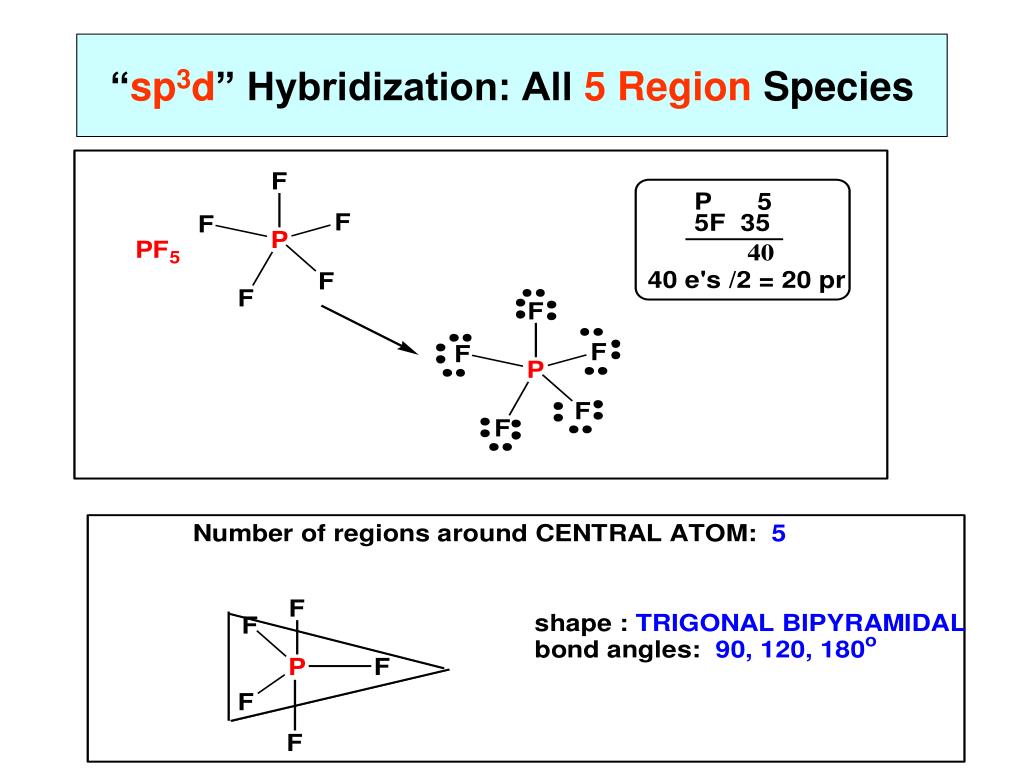
Valence bond theory would predict that the two O–H bonds form from the overlap of these two 2 p orbitals with the 1 s orbitals of the hydrogen atoms. Oxygen has the electron configuration 1 s 22 s 22 p 4, with two unpaired electrons (one in each of the two 2p orbitals). As an example, let us consider the water molecule, in which we have one oxygen atom bonding to two hydrogen atoms. 
However, to understand how molecules with more than two atoms form stable bonds, we require a more detailed model. Thinking in terms of overlapping atomic orbitals is one way for us to explain how chemical bonds form in diatomic molecules. This is not consistent with experimental evidence. The hypothetical overlap of two of the 2 p orbitals on an oxygen atom (red) with the 1s orbitals of two hydrogen atoms (blue) would produce a bond angle of 90°.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
